Within seven weeks of its fast COVID-19 tests gaining approval from Australian health authorities, Sydney-based Atomo Diagnostics (ASX: AT1) expects to take its technology to one of the countries worst-hit by the pandemic.
Atomo has entered into an agreement with PinkTech Design Private Limited, trading as DIVOC Laboratories, to launch its AtomoRapid COVID-19 antibody test in the Indian market.
AtomoRapid has been approved by Australia's Therapeutic Goods Administration (TGA) and has the European Union's CE marking, but before it enters the Indian market it will still need to be approved by the country's authorities.
The company explains DIVOC expects to obtain product registration approval for professional use in the second quarter of FY21.
DIVOC sees potential for the product's distribution to government, corporate and lab-to-lab settings, as well as through an established home visit network for testing that is supported by medical professionals.
Under the deal, Atomo will initially provide 77,000 antibody test kits with one per container, and the agreement will terminate if DIVOC fails to order one million units over the following 12 months following receipt of regulatory approval in India.
In return, Atomo will receive a fixed transfer price per unit and will also receive a percentage of revenues received by DIVOC on final product sales above this transfer price.
Globally, India is currently experiencing the highest rate of daily infections, with the seven-day average currently exceeding 90,000 daily cases.
The South Asian nation has been the second-worst-hit in terms of total recorded cases at more than 5.6 million, of which almost one million are still active.
India's testing rate per capita is a fourth of the US and its death rate per capita is similar, amounting to more than 90,000 lives taken to date.
The right to the non-exclusive sale of the AtomoRapid COVID-19 rapid test in India is a further expansion of Atomo's agreement with NG Biotech, its French partner that manufactures the device's strip while the product itself is made by the Australian company.
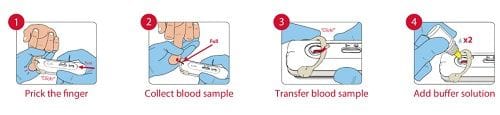
The device uses Atomo's locally-developed 'Galileo integrated diagnostic test platform, which has been in use for HIV self-testing and is also TGA-approved for that purpose.
"We are delighted to be able to offer our antibody rapid test in another large international market," says Atomo Diagnostics co-founder and managing director John Kelly.
"Rapid testing forms a significant pillar of India's response to managing the COVID-19 pandemic with the numbers of daily rapid tests increasing significantly in recent months.
"Our Indian partner is a high-quality provider of diagnostic services, being one of the few laboratories in India that has been able to secure NABL [Indian National Accreditation Board for Testing and Calibration Laboratories] accreditation.
"We are confident of their ability to rollout the AtomoRapid COVID-19 antibody testing across a number of high value channels in India in the coming months."
AT1 shares rose 7.14 per cent today to $0.375 each.
Updated at 4:13pm AEST on 23 September 2020.

)
)

