Melbourne-headquartered nuclear medicine company Telix Pharmaceuticals (ASX: TLX) has completed a $175 million institutional placement to fund final-stage trials of a prostate cancer therapy and the roll-out of its first commercial product Illucix, a prostate cancer imaging product approved by the US Food and Drug Administration (FDA) in December.
The new funds were raised at $7.70 per share and the company has also launched a $25 million share purchase plan (SPP) at the same price, although TLX shares plunged well below that level today, perhaps as investors got spooked by co-founders and leading shareholders Dr Christian Behrenbruch and Dr Andreas Kluge agreeing to sell 8.1 per cent of their respective holdings.
Cash reserves were also cut in half in the last three months of 2021, falling from $45.85 million to $22.04 million, but this was in line with forecasts. Out of the total operating outflows almost $7 million was tied up in R&D spending.
Nonetheless, managing director and CEO Dr Behrenbruch was upbeat during the announcement that follows the approval of Illuccix on 20 December, tapping in to a distribution network of more than 140 nuclear pharmacies through Telix's agreements with Cardinal Health and PharmaLogic in the US.
"We are delighted with the level of support for the placement from new and existing investors," he says.
"Funds raised under the placement and SPP will provide the company with the financial resources to execute on the next phase of Telix’s growth strategy, which is to advance our core therapeutic clinical programs, expand our commercial diagnostic portfolio and proactively seek out new innovations that will cement Telix’s position as a leader in the field of ‘theranostic’ radiopharmaceuticals."
The company has clarified this is the first sale of shares by Dr Behrenbruch or Dr Kluge since Telix's inception in 2015, and the pair have pledged they will not sell any further shares in Telix at any time in the 12 months following settlement.
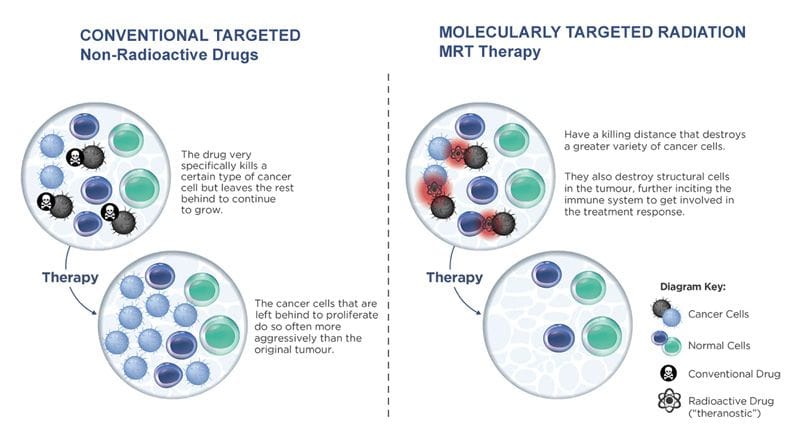
With the extra cash, the molecularly targeted radiation (MTR) company plans to place a particular focus on its ProstACT (prostate cancer therapy) clinical program, and further expand its pipeline and capabilities around Targeted Alpha Therapy (TAT).

Telix’s lead prostate cancer imaging product Illucix has now received regulatory approval in both the US and Australia, where roll-outs are on the cards in the current quarter, while the product was also granted an exceptional authorisation from the Brazilian Health Regulatory Agency allowing it to be marketed and sold in Brazil ahead of full regulatory approval which is anticipated to be received in 2022.
"To achieve our first regulatory approval– in both the United States and Australia – is a major achievement and marks our transition to a commercial-stage company," the CEO says.
"In the initial stages of launch we will focus on our existing users and possible early adopters to build a foundational customer base, as well as executing a wide-reaching market access and education campaign to nurture new customer sites and create demand, ahead of securing reimbursement status for Illuccix in the United States.
"The validation of our first regulatory approvals, the depth and quality of our clinical pipeline and our strength in supply chain and manufacturing uniquely positions Telix as a vertically-integrated leader in the global radiopharmaceutical industry. We look forward to progressing on all fronts in 2022 and further unlocking the value inherent in our pipeline."
In preparation for an approval decision notification and commercial launch, Telix has also continued to build out its distribution network in Europe. In the December quarter the group entered into an exclusive three-year commercial distribution agreement with Nucliber in Spain, and in early 2022 signed a three-year agreement with Biokosmos for exclusive distribution rights in Greece and Cyprus. These add to existing partnerships already in place for other major European markets.
Still in the early stages of its commercialisation plans, Telix only recorded $0.99 million in receipts from customers in the December quarter, but its share price has soared from $3.99 at the start of 2021 to $8.80 at the start of 2022. Today the share price has pulled back down by 14.4 per cent to $6.925 at the time of writing.
Get our daily business news
Sign up to our free email news updates.

)
)
)
)
)
)

